Software for Medical & Regulated Systems
We design and build medical and regulated software where reliability, auditability, security, and long-term support are non-negotiable. Because excellence in medical device systems matters.
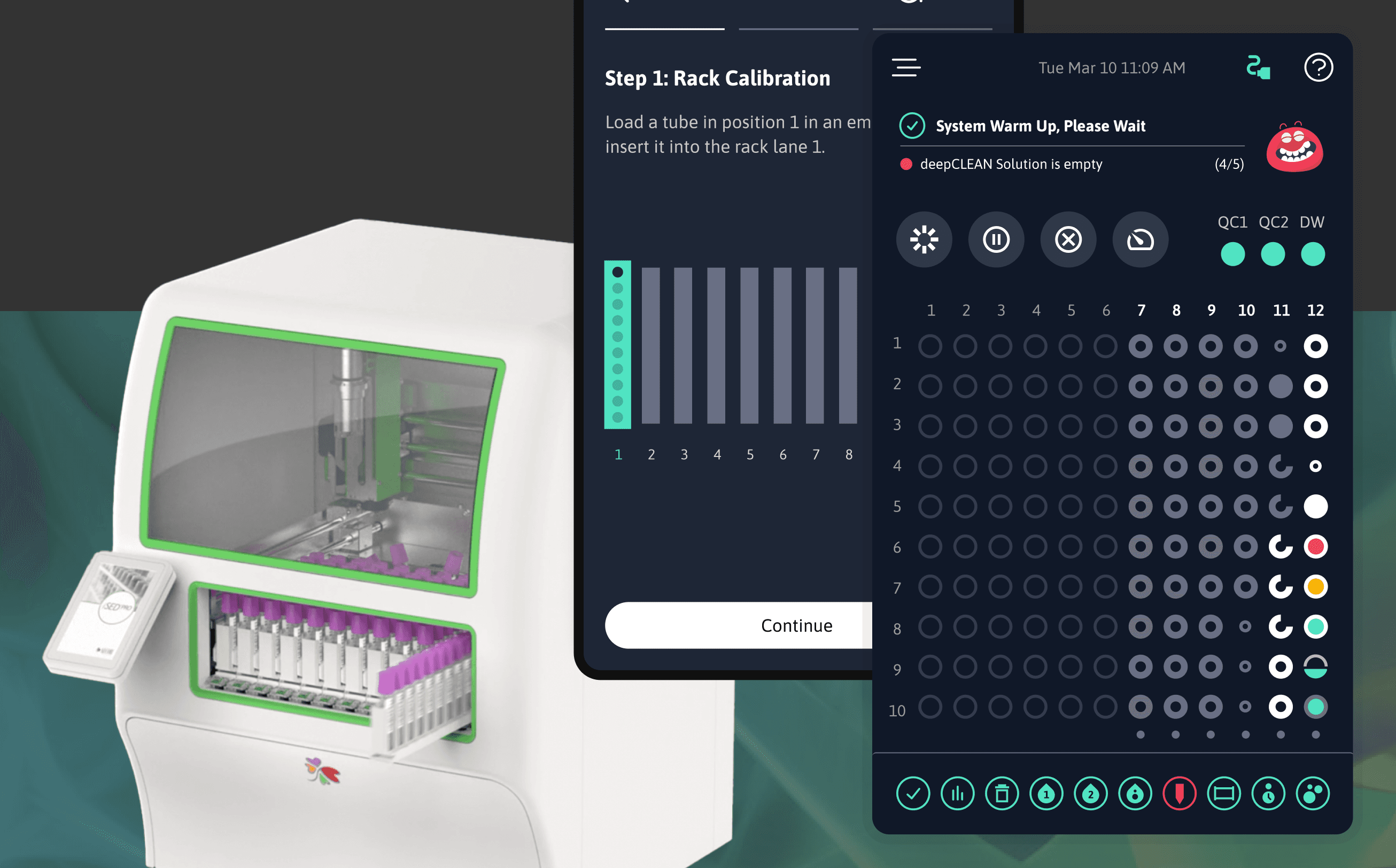
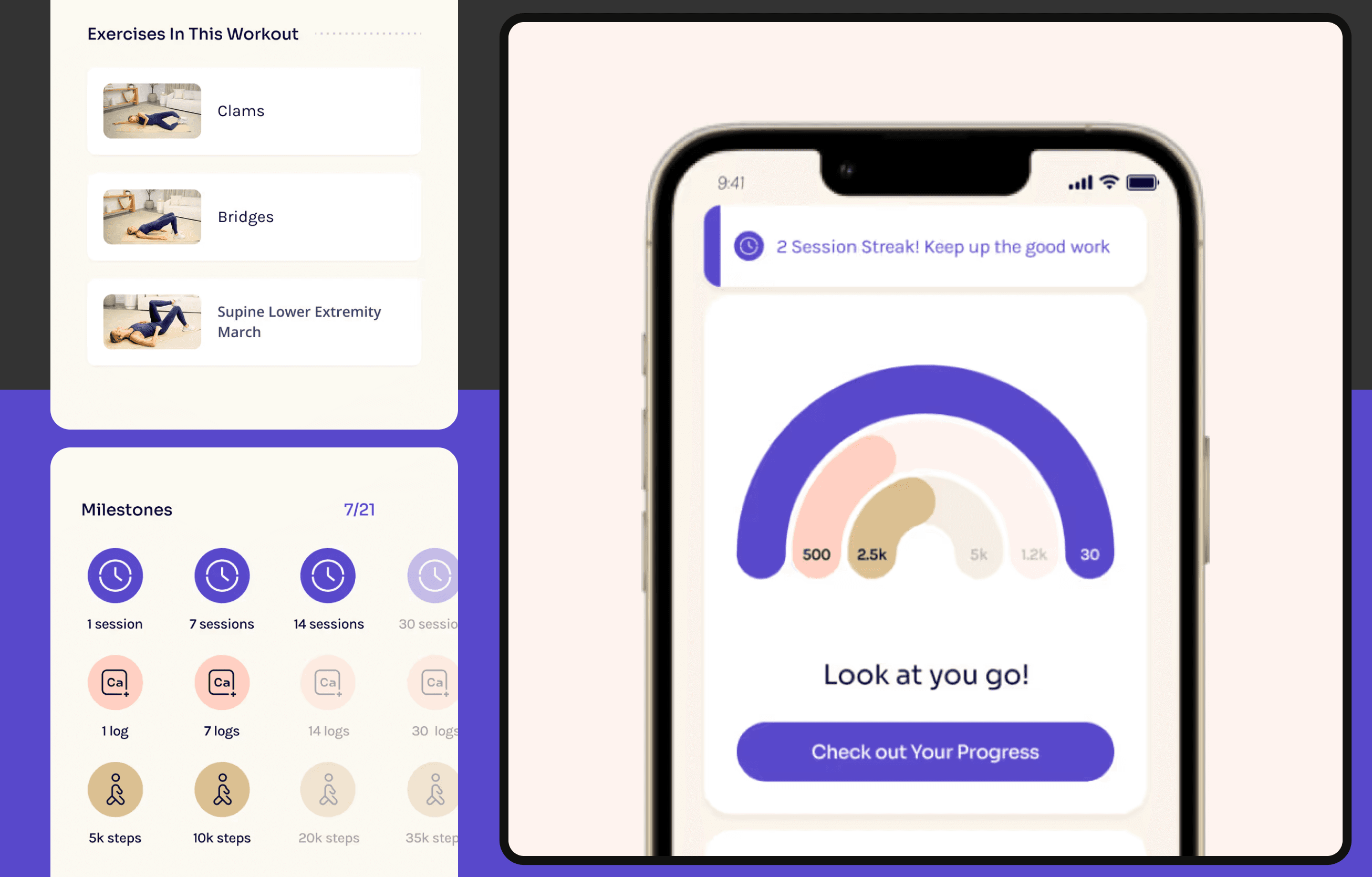
See All Medical Work →Developing software for medical and wearable devices is tricky.
It requires a deep understanding of the capabilities of the hardware. Knowledge of how to handle the interplay between the hardware, the software, and the cloud. Nuances around battery life, compute, latency, offline.
Perhaps most importantly, it requires an understanding of the documentation, processes and procedures necessary for regulatory compliance and FDA certification. You don’t want to wing it. You want to partner with a firm who’s been there before. Township is that firm.
We understand hardware.
Our software engineers are familiar with the capabilities (and limitations) of medical hardware. We work side-by-side with the hardware team to rigorously test and design solutions that balance performance and reliability.
Future proof your solution.
It’s easy to ship software. Hardware is much harder. Even firmware has its risks. Township helps clients anticipate future needs so they ship the right product the first time.
FDA Compliance.
Township provides detailed documentation and support to help you meet stringent FDA regulations, reducing the risk of compliance issues and time delays.
Ready to build something that matters?
We’re especially helpful when the path forward isn’t obvious yet. Let’s discuss your challenge and map out the smartest path forward.
Other Services
Growth Systems →
Marketing sites that do way more than a landing page. Sites that integrate with your business tools, build trust with your audience, and scale with your growth.
Connected & Intelligent Systems →
Systems that connect, adapt, and improve over time. We connect modern user interfaces, automation, data, and AI where they meaningfully improve outcomes.